Same volume, same weight?
2. Compare the weights

Introduce the electronic scales
Distribute the electronic scales that students will be using in this unit. Explain that that scales have been made specifically for weighing objects that are light. They could weigh a couple of apples, but not a brick. The class will be using just light objects in the science unit. Students should handle the scales carefully and not press down on them or place any heavy objects on them.
Letter from the Engineer
A better way to weigh
This scale you are about to use looks very different than the pan balances you may have used in the past to weigh things. Do you remember how to weigh things using a double pan balance? You place an object you want to weigh in one pan and add weights to the pan on the opposite side until the two pans are level.
Engineers know that a pan balance is difficult to use. Sometimes the weight you add is too heavy or too light and nothing seems to make the pans balance perfectly. Sometimes the weight you need can get lost. And even when you can balance the pans perfectly, it can take a lot of time. Engineers saw these problems and asked, “How can we improve this instrument? Can we make a scale that is easier and faster to use, is accurate, and that does not need a set of weights to balance an object. One solution they came up with is the electronic scale, which is the kind of scale you are about to use.
How does the electronic scale work? It depends on the fact that placing weight on something causes it to bend or change its shape. Sometimes you can see this very clearly: Think about a diving board. Once someone walks out to the end of the diving board, it bends to a new position. The change in shape or position is called a “deflection”. After the person leaves the diving board it returns to its original position and the deflection that was caused by the weight goes away.
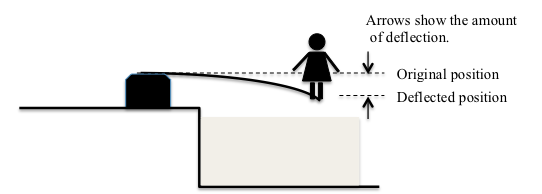
Even when the weight is very small, deflection happens, although it can be too small for your eyes to notice. If a small bird landed on the end of the diving board, the board would deflect a very tiny amount but no one would notice it.
When we place an object on the top of an electronic scale, we are causing a small piece of metal inside the scale — you can think of it as a tiny diving board — to bend or deflect; the heavier the object, the more the piece of metal deflects. Engineers had to study how different materials deflect or bend when weight is put on them; some deflect much more than others. They have designed a way to measure that deflection, and to have batteries send a certain amount of electricity to the LCD (screen that shows the weight) depending on how much the metal deflects. More deflection means more weight. Both the pan balance and the electronic scale are examples of technology that help us to weigh things, and the electronic scale is a good example of how engineers have developed a new technology to solve a problem.
Demonstrate how to turn the scale on and have students turn on their scales.
Some scales can be set to measure either in grams or in ounces. For all work in this unit, students should check the units each time before they start to use the scale, and use the switch or button to reset it to grams if that is necessary.
Finally, a scale will sometimes indicate a weight, or even a minus number, when nothing is on it. This would result in an incorrect measurement. Show students how to reset the scale to zero; they should always be sure the scale reads zero before they weight anything.
Have students place a pencil or other small object on the scale and observe the way in which the scale displays the weight.
Compare two liquids
Distribute a tray of materials to each group and identify the four earth materials: water, mineral oil, organic soil, and sand.
Hold up the containers of water and mineral oil and confirm that they hold the same volume of liquid. It is not necessary to measure the volumes, just ask the class to compare them "by eye."
Before you weigh the two samples, make a prediction: How do you think the weights compare?
Listen carefully to the reasoning. Do students think the weights will be the same “because the volumes are the same”? Do they think they‘ll weigh the same “because both materials are liquids”? Or maybe they think the weights will be different because the liquids have different properties, perhaps “because one liquid is thick and the other is thin.” Acknowledge all answers and theories.
- How can we find out for sure?
- By weighing the samples
If students don't raise the question themselves, ask how they can measure the weight of only the liquid
How can we weigh just the water, or just the mineral oil?
Listen for a suggestion to subtract the weight of the empty container from the total weight of the sample. Introduce the term tare weight, which is the weight of just the container.
Have groups weigh an empty container and record that weight (approx. 26g) in all four rows of the data table their Science Notebooks [Observations of weights and volumes]. Next have them weigh the two liquid samples, calculate the weight of just the liquids, and enter their data in their notebooks. Make sure they specify that the unit of weight is grams.
When all the groups are finished, ask for their results and record them in the table you prepared before class. Do not try to reconcile differences at this point.
Example of a class data table.
| Material | Volume or Bulk Volume |
Weight (g) Material |
Class Agreement Weight (g) |
|---|---|---|---|
| Water (liquid) |
40cc | 40g; 40g; 39; 40g; 41g; 39g | |
| Mineral Oil (liquid) |
40cc | 32g; 31g; 31g; 32g; 33g; 33g | |
| Sand (granular solid) |
40cc | 78g; 76g; 77g; 77g; 78g; 78g | |
| Organic (granular solid) |
40cc | 17g; 16g; 15g; 16g; 16g; 20g |
Compare two solids
Have students pass around the containers of sand and organic soil.
- Do the samples seem to have the same volume?
- Do you think they have the same weight?
Have students weigh the samples as before, subtracting the tare weight and recording the data in their notebooks. Make sure they label their measurements in grams.
The procedure is exactly the same as before, but this time there’s a wrinkle — there are empty spaces between the grains of soil.
- Are the empty spaces parts of the volume?
Yes. Sand and organic soil belong to a group of materials called granular materials. Cheerios, sugar, gravel, and peanuts are also granular materials. Granular materials are made up of many smaller pieces that are about the same size and that have spaces in between them. The volume of a granular material is the sum of two smaller volumes: 1) the volume of all the individual pieces, and 2) the volume of all the small spaces. This combined volume has a special name: it is called bulk volume. Bulk volume is measured the same way that the volume of a liquid is measured, but the name bulk volume lets everyone know that the volume is a combination of the individual pieces and the spaces between them.
Once students have weighed the samples and entered the data in their notebooks, enter their data in the class table.
Allow time for students to briefly discuss the reflection questions in their small groups and write responses in their notebooks before moving on to the class discussion [Reflections on weights and volumes].