What does a drop of water weigh?
Plan Investigation 4

In the previous investigation, students found evidence that 1cc of water weighs 1 gram.
How would we measure the weight of a single drop of water? When we put a drop of water on our classroom scale, it doesn't register any weight at all.
In this session students use a small dropper and a basic understanding of fractions to figure out the weight of a single drop of water. Why is this important? This starts a sequence of investigations in which students extend their understanding of what it means for an object to have weight. They progress from experience with objects that have clearly perceptible weight to objects for which weight is barely perceptible, such as a single drop of water. Eventually they consider particles too small to see, whose weight becomes apparent only when billions of them are present.
By the end of this investigation students will understand that when a sample of material is too small to weigh on their scales, they can use their knowledge of unit weight and volume to calculate the weight. They will also deepen their understanding that even the tiniest objects, such as a single grain of sand, have weight.
Learning Goals
- Understand that very small things have weight
| Sequence of experiences | ||
|---|---|---|
| 1. Ask the question | All Class | 5 Mins |
| 2. Develop a strategy | All Class | 10 Mins |
| 3. Explore | Individual | 15 Mins |
| 4. Share the results | All Class | 5 Mins |
| 5. Make meaning | All Class | 10 Mins |
NGSS Alignment
 Three Dimensional Learning
Three Dimensional Learning
| Disciplinary Core Ideas | 5-PS1-1, 5-PS1-2
Materials have weight and take up space and are matter. Very small pieces (particles) of matter have weight and take up space. |
| Practices |
Engaging in argument from evidence: : In the last investigation, students learned that 1cc of water weighs 1g. They count the number of drops of water released from a 1cc syringe and, using mathematics, they make claims about the weight of a single water drop. Constructing explanations: : given that a single water drop or grain of sand has no perceptible weight, students explain why a bucket of water or pail of sand can have significant weight. |
| Crosscutting Concepts | Scale, proportion and quantity: students apply their knowledge of the weight:volume ratio of water to establish the weight of a single drop of water. |
Materials and Preparation
For the class:

- Post the investigation question in a place where all students can see it.
- Make a class table titled, "What does a drop of water weigh?"; an example is found in Step 4.
- 1 digital scale
For each group:
- 2 12oz cups approximately 1/2 full of water
- 4 1cc droppers (1cc small syringe)
- 4 centimeter cubes
- 4 paper towels
Notebook Pages
1. Ask the question
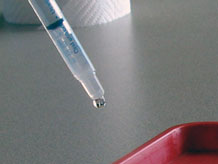
We know how to weigh the amount of water in our mini-lakes, but how could we weigh the water in a raindrop? Introduce today's investigation question:
What does a drop of water weigh?
Give students a chance to share ideas. Challenge students to help refine the investigation question:
- Are all drops the same size?
- What size drop of water do we weigh?
- Can we control the size of a drop?
- Can we rely on the classroom scale to provide the answer?
Acknowledge that drops can be different sizes, and explain that the class has a tool-a dropper-that makes a uniform drop of a certain size.
Show students the dropper, a 1cc syringe. The class will address today's investigation question by determining the weight of one drop from this dropper.
Revise the investigation question to read:
What does a drop of water from our classroom dropper weigh?
Post the rephrased investigation question.
2. Develop a strategy
Strategy 1: Use the scale.
It's likely someone will suggest putting one or more drops on a scale. Explain that if we had more accurate scales, this strategy could work.
Demonstrate placing a single drop of water on the scale. The scale will not register any weight. Even when placing multiple drops of water on the scale, one drop at a time, it's unlikely these scales will register weight. If they did, the weight would be rounded to the nearest gram, providing inaccurate results.
Encourage students to come up with another strategy.
Strategy 2: Use the information that 1 cubic centimeter of water weighs 1 gram.
- There is a way to use something we discovered in the last investigation-that one cubic centimeter of water weighs one gram-to help us answer the investigation question. How might counting the drops in 1cc of water help us?
- If we could make just 2 drops of water from one whole cubic centimeter of water, what would each of those drops weigh?
- If we could make 10 drops of water from one whole cubic centimeter of water, what would each of those drops weigh?
Fill and weigh a dropper
Place one empty dropper on a classroom scale and read the weight (approximately 3g).
Point out that the dropper has marks on it that allow us to add exactly one cubic centimeter of water. Demonstrate a standard procedure for accurately filling the dropper with 1cc of water:

- Push the plunger as far as it will go into the barrel of the dropper.
- Put the tip of the dropper in a cup of water and slowly pull on the plunger to completely fill the dropper.
- Point the tip of the dropper towards the ceiling and tap the barrel with a finger until all air bubbles have risen out of the water.
- Carefully depress the plunger until the black ring of the seal aligns with the 1cc mark.
Place the dropper on the scale again. It should weigh one gram more than its weight when empty.
Demonstrate making drops

To answer the question, What does a drop of water from our classroom dropper weigh?, it's important that everyone use the same tool in the same way in order to get accurate results. Demonstrate a standard procedure to make drops:
- Hold the dropper vertically, tip down, about 1 inch above a desk top.
- Gently push the plunger to release one drop of water.
- Move the dropper sideways about 2 inches and release another drop.
- Form a line of 5 or 6 drops.
Keeping the drops separate from one another makes it possible to count them after the dropper is empty, in the event someone loses count while making the drops.
3. Explore

Distribute the materials. Students use the paper towel for drying desktops after counting the drops. The purpose of the centimeter cube is to show the volume of the 1cc dropper in another shape - to reinforce the size of 1cc of water. Each cubic centimeter takes up the same amount of space, as the one cubic centimeter of water students will add to their droppers.
Emphasize the importance of everyone using standard procedure for both filling and emptying the droppers. Ask students to practice once before they use the droppers to get a final count.
Students should make a long row of drops on their desktops as they slowly empty the dropper, keeping the tip about 1 inch above the desktop.
Note: Since the droppers can become a distraction, collect them as soon as students have completed their actual count.
4. Share the results
Survey the class for the results and enter their data in a class chart, asking each student to determine the weight of one drop once they provide the number of drops made.
| Weight of water | Number of drops | Weight of 1 drop |
|---|---|---|
| 1g | 29 | 1/29thg |
| 1g | 30 | 1/30thg |
| 1g | 32 | 1/32ndg |
| etc. |
Address the investigation question:
What does a drop of water from our classroom dropper weigh? Does a drop of water weigh more or less than one gram?
- approximately 1/30th of a gram?
Have students complete the Science Notebook page [Reflecting on the weight of small bits of matter].
Note: Here is another example of small differences in a situation where one might expect everyone to get the same result. Students should understand the possible reasons for the differences and agree to select a "typical" value for the weight of one drop.
5. Make meaning
Acknowledge that students have just finished some interesting work; they used mathematics to figure out the weight of something that had too little matter for the classroom scale to sense.
Purpose of the discussion
The purpose of this discussion is for students to jointly construct explanations for how objects such as a bucket of water or sand can have significant weight, given that a single drop of water or grain of sand has no perceptible weight.
Engage students in the focus question
If a drop of water weighs just 1/30th of a gram, how do you explain why a bucket of water is so heavy?
- There are many, many drops of water in a bucket full of water.
- Each drop contributes something to the total weight of the bucket of water.
Allow time for students to formulate responses. Encourage full participation by asking for others to share their own explanations and to respond to the explanations of others.
Do you think a single grain of sand weighs anything? Explain your answer.
- If students claim "yes", what reasoning can they provide to support their claim?
- If students claim "no", ask for the explanation; it will help you determine whether students are focusing on whether a grain of sand will register any weight on the scale, or if students believe a grain of sand has no weight.
How does a bucket of sand get to be so heavy when a grain of sand weighs so little?
- There are many millions of grains of sand in a bucket, with each grain contributing a tiny bit of weight to the total weight of the bucket of sand.
Can you think of other examples of tiny bits that seem to have no weight individually, but that can be combined together to have significant weight?
- Grains of sugar or salt; particles of flour.
Summarize the discussion and recap the investigation
Using the same language students have used, summarize their main ideas. Include the following key ideas:
- If a large collection of grains of sand (or drops of water, etc.) have weight, then we can reason that each individual grain must have some weight, even if that weight is too small to sense or measure.
- Although individual grains of sand or drops of water may have imperceptible weight, combining millions or billions of them together can result in significant weight.
Recap the following:
- Tiny things, such as drops of water, are not heavy enough to register weight on the classroom scales.
- We've figured out how to calculate the weight of a water drop and concluded that tiny things such as single drops of water or grains of sand actually have weight.
When we think about how heavy a bucket of water feels, or a bucket of sand feels, it can make us wonder, "How many of those drops, or grains, are there in a bucket, to make it feel so heavy?"